Beryllium has a +2 oxidation allowsįorming many covalent dissolves in non- oxidizing acids. While bonding with the other atoms, it has a very high ionization In the air, beryllium resists oxidation and is extracted by the These characteristics make unique stability under thermal loading. Very high melting point which makes beryllium metal the best heat dissipationĬharacteristics per unit has a low coefficient of linear thermalĮxpansion. It has a close-packed hexagonal crystal has a Emerald is known to be a variety of beryl. Friedrich Wohler at berlin in 1828 isolated the beryllium metal.īeryllium generally occurs in the earth’s crust to the extent of 0.0002 
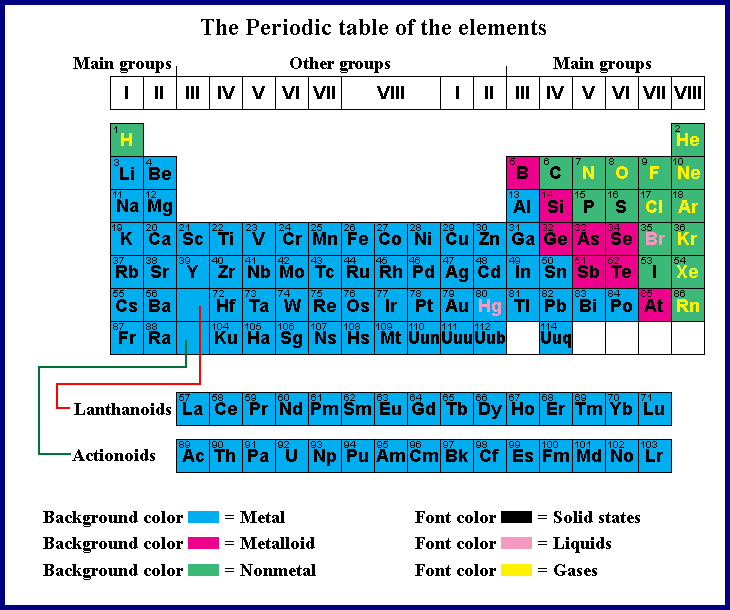
Beryllium was discovered inġ798 by French chemist Nicolas-Louis Vauquelin. Of alkali metals are often insoluble in water ALKALINE METALSġ metals are less reactive than group 2 metals. Group 2 elements are called alkaline metals because they formĪlkaline solutions, hydroxides, when reacting with water and their oxides areįound in the earth’s crust. GROUP 2 OF THE PERIODIC TABLE Group 2 elements are: Br To show patterns in atomic radius, ionization energy, and electronegativity. Same group elements have similar properties and reactivity. Each group of elements having the same number of valence electrons. S-, p- and d-block elements of the periodic table are arranged into these columns The 18 vertical columns of the table are called Groups. There are also more specific groups like alkali metals, transition metals, rare metals, alkaline earth, halogens, and noble gases. There are multiple ways of grouping the elements but they are commonly divided into metals, non-metals, metalloids. In the periodic table of chemical elements, there is a group (also known as a family) is a column of elements. 
Side of the periodic table GROUPING METHOD Metals are on the left side and non-metal are on the right Horizontally in order of increasing atomic number and vertically according to In the periodic table, chemical elements are present in rows The seven rows of the table are called a periodic table. * Some caveats: for example, aluminium will react slowly with water, if the thin aluminium oxide layer that prevents it from reacting is damaged.It is a tabular display of chemical elements, arranged in order of atomic number in rows so that elements with similar atomic structure and recurring chemical properties appear in vertical columns. * This graphic doesn’t contain every metal in the periodic table. Majority of metals occur naturally in compounds, which we must remove them from. Some metals are so unreactive they occur largely uncombined with other elements, simple to obtain. This is because they can react with the compounds in metal ores, and displace the metals, aiding with their extraction. Carbon and hydrogen are also shoehorned in between entries in the list, despite being non-metals. Copper sulfate + zinc → zinc sulfate + copper Magnesium sulfate + zinc → NO REACTION The reactivity series also gives us an insight into why different metals are extracted from their ores in different ways. Conversely, if we react a metal with another metal lower in the series, no reaction will take place. The more reactive metal will take the place of the less reactive metal in the compound. If a metal compound reacts with a metal that’s above it in the reactivity series, a displacement reaction will occur. Uses of this: * predict the outcome of certain chemical reactions. Transition metals are much less reactive Gold and platinum have little in the way of chemical reaction. Closely followed by the marginally less reactive group 2 metals. Metals have a range of reactivities: Videos: the classic alkali metals in water demonstration The reactivity series offers a ranking of the metals in order of their reactivity. The text below has been excerpted from Compound Chemistry Student handout: Reactivity Series of Metals (PDF) This graphic places a selection of common metals into order of reactivity, as well as showing their reactions with air, water and steam. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
